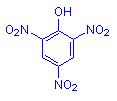
Trinitrophenol
Description
Picric acid was the first synthetic dye to be made, in 1771 by Woulfe. It is now most commonly used in conjunction with acid fuchsin in the Van Gieson method which demonstrates collagen (red) in contrast to cytoplasm and muscle (yellow). It is also used, with or without other yellow acid dyes, to colour erythrocytes in trichrome methods for fibrin such as Lendrum's Picro-Mallory. It is a fixing agent and is a constituent of Bouin's fluid. It has been used as a differentiating acid in a variant of Heidenhain's iron hematoxylin, and is a trapping agent for methyl violet in the Gram Weigert stain for bacteria and fibrin.
Picric acid is often used in conjunction with acid fuchsin to differentiate cytoplasm and muscle from and collagen. Such techniques are referred to as picro-fuchsin staining, and numerous formulations or variants exist.
References
- R. D. Lillie.
Conn’s Biological Stains
Williams & Wilkins, Baltimore, MD., U.S.A. - Edward Gurr, 1971
Synthetic dyes in biology, medicine and chemistry
Academic Press, London, England. - Pope Sine,
Synthetic dyes
Rajat Publications, New Delhi, UK.





