The Hematoxylin and Eosin stain (H & E) may well be the most widely used histological staining method of all. It may also be the most poorly done, and the most poorly understood. Certainly, the quality varies considerably from laboratory to laboratory. This is unfortunate, as a well stained H & E section can yield a surprising amount of information.
Example H&E Stains
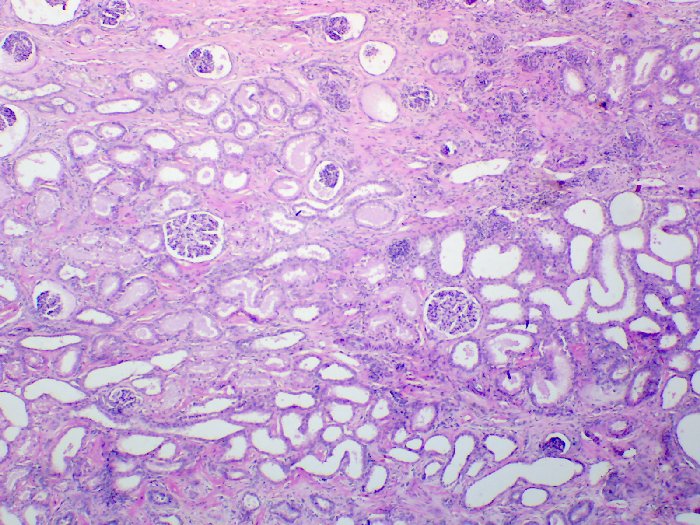
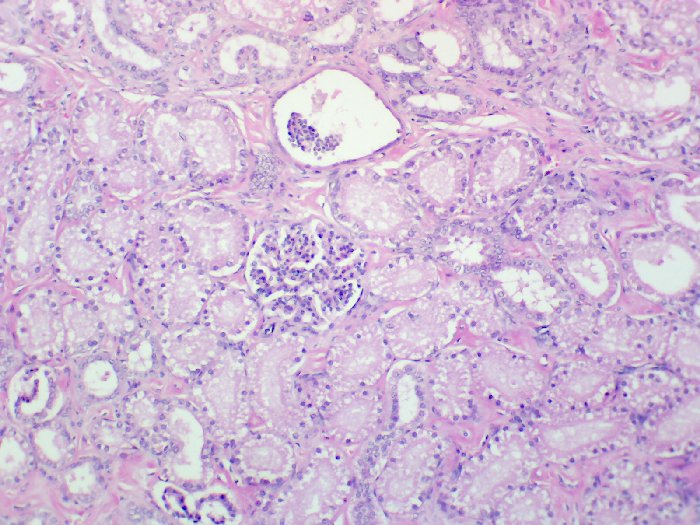
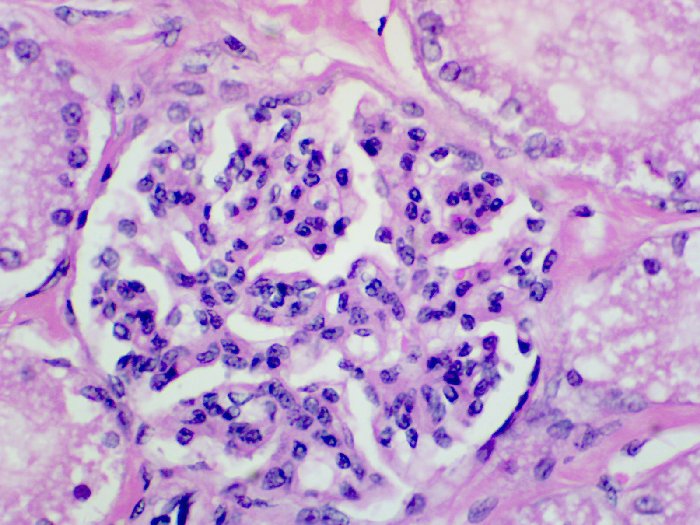
The following images depict human kidney stained progressively with hemalum and eosin Y at 40x, 100x, and 400x magnification, respectively.
Hematoxylin
The H&E stain requires two histological stains, with hematoxylin as the first component. There are many considerations to selecting and formulating the proper solution, from the hematoxylin type, acid content, staining time, blueing, and more. Learn more from the resources below.
Eosin
The second component of the H & E is eosin, the counterstain. Similarly, considerations around the solution type, use of acetic acid, and selection of dyes can impact the final resulting appearance. Learn more from the detailed guide below.
Considerations for H&E Staining
Any high-quality histological preparation is dependent on the initial handling. Poorly fixed and processed tissue will never be stained optimally. Unfortunately, modern constraints in a clinical setting, where the emphasis is on rapid diagnosis and high volume, often preclude using those procedures which lead to high-quality preparations. In other words, the modern penchant for rapid fixation and processing works against high-quality H & E staining. This is not a question of competence, it is a limitation due to the processes used.
A few hours in 10% neutral buffered formalin is inadequate for the proteins to properly react with the fixative. Similarly, the practice of deliberate under-dehydration as a means of masking inadequate fixation should be avoided if high-quality staining is to be achieved. Ethanol is a fixative, and if the initial fixation is inadequate, the ethanol will continue to fix the tissue while dehydrating. Since ethanol is a poor fixative when used alone, tissues will be brittle and shrunken, with a tendency to shatter during sectioning and will have poorer staining. It would be far better to change to a rapid fixative and apply long enough for it to complete its work, then dehydrate properly before clearing and infiltrating.
A further qualification must also be made. What constitutes a “good” H & E differs from person to person. This is so partly due to application – an anatomist might be interested purely in a clean illustration of structure, for instance, and appreciate blue nuclei with clean pink background, whereas a pathologist may be interested in seeing cellular RNA darkly stained as an aid to diagnosis. There may also be preference differences within the same institution between pathologists. There is no right or wrong about this. Preferences do vary, and it is the obligation of the histotechnologist to stain preparations consistently to the criteria preferred.
Rather than arbitrarily saying a particular appearance constitutes a “good” H & E or how to achieve the “best” staining, this article will focus on how to achieve the staining appearance desired. In other words, it will, hopefully, help in understanding how to modify the staining and how to get it to look a certain way.
This does raise a point, though. The histotechnologist should be aware of what appearance the viewer wants. Often this has never been specified. Often the appearance aimed for has arisen by a process of selection, slides being rejected, restained, or criticized, for instance. Only rarely does the histotechnologist stain a series of adjacent sections to show progressively different appearances and have the final viewer select which appearance is most useful. It is strongly recommended this be done periodically. The placenta is a suitable tissue.
Protocols
Hematoxylin and Eosin Staining Protocols
Find step-by-step instructions for the gold standard stain in histology, the H & E stain.
References
- Gray, Peter. (1954)
The Microtomist’s Formulary and Guide.
Originally published by: The Blakiston Co.
Republished by: Robert E. Krieger Publishing Co. - Horobin R W & Kiernan J A. (2002)
Conn’s Biological Stains, 10th ed.
BIOS Scientific Publishers, Oxford, UK - Susan Budavari, Editor. (1996)
The Merck Index, Ed. 12
Merck & Co., Inc., Whitehouse Station, NJ, USA - R. H. M. J. Lemmens and N. Wulijarna-Soetjipto, Editors. (1992)
Plant resources of South East Asia No. 3, Dye and tannin-producing plants
PROSEA, Bogor, Indonesia. - Morsingh, F., and Robinson, R. (1970)
The synthesis of brazilin and haematoxylin
Tetrahedron,v.26,p.281,
UK - James Cook University, School of Marine and Tropical Biology
Discover Nature
Townsville, Queensland, Australia