There are two kinds of dipole-dipole interactions. The stronger of the two is the hydrogen bond. The weaker is the van der Waal’s forces. Both interactions depend on the same fundamental cause, the charge on electrons, and how that results in attraction and repulsion at an atomic level.
van der Waal’s Forces
The structure of an atom has a central nucleus composed of positively charged protons and uncharged neutrons surrounded by a varying number of negatively charged electrons. The electrons orbit the nucleus. As they do so, there are minor variations in the expression of their charge. At the point around the atom that the electron occupies at that instant, expression of its negative charge is increased, and at the point opposite to that, where its influence is the least, the expression of the proton’s positive charge is likewise increased.
Of course, we are not dealing with two individual atoms. Billions are involved in any interaction between compounds. At any one time, there will be numerous instances of molecules that have a temporary charge distribution that results in an attraction for another molecule. Single instances are not important. Very large numbers of instances of such very weak and fleeting attractions, however, can result in substantial bonding between the compounds. It is the large number of instances involved that is important, not the nature of the attraction.

Two atoms interacting due to the influence of their electrons (the red dots). One is shown with a stationary electron to illustrate how its position relative to both the electrons and the protons of the other atom affect the relationship between the two.
Hydrogen Bonds
Hydrogen bonds are somewhat stronger than van der Waal’s forces, and require two components: a donor group and an acceptor group. The donor group is composed of hydrogen and an electronegative atom. The acceptor group is another electronegative atom that has available electrons.
When hydrogen and another electronegative element combine in a covalent bond, the electrons in the resulting molecule may be attracted away from the hydrogen to the other element and locally increase its negative charge. The hydrogen atom develops a corresponding positive charge. The charges around the molecule are uneven and one part has a greater negative charge and the other has a greater positive charge. Molecules such as this are termed polar compounds. The elements are commonly oxygen, nitrogen, sulphur, halogens or carbon.

The picture to the left illustrates the polarity of a water molecule. The electron from each of the hydrogen atoms (yellow) is attached to and shared by the oxygen atom (light blue). Due to the significant difference in the electronegativities of the two atoms, the electrons are attracted more to the oxygen than to the hydrogen and tend to be displaced, more or less permanently, away from the hydrogen atom. The end result is a molecule positively charged on one side (hydrogen), and negatively charged on the other (oxygen).
Due to their polarity, many hydrogen bonding donors are also acceptors, but this is not always necessarily so. The common molecules we deal with in histotechnology frequently do contain both donors and acceptors. These molecules include water, alcohols, phenols, carboxylic acids and amines. Some compounds do not act as donors or acceptors, and these include some saturated hydrocarbons. The strength of an individual hydrogen bond is variable because the electronegativities of the particular element to which the hydrogen is attached can vary. This results in differing degrees of polarity depending on the particular atoms involved. We can sometimes use these differences in the degree of polarity to control the staining process. An example would be the use of ethanol as a solvent instead of water in amyloid staining with congo red.
The basic principle behind hydrogen bonding is that the electron deficient hydrogen atom of one polar molecule is attracted to the electron rich side of another polar molecule. An electron deficient hydrogen can also be attracted to an electron rich atom in the same molecule in some compounds such as polypeptides. While the displacement of electrons in polar molecules is essentially permanent and the attraction of proton rich hydrogen for electron rich atoms is similarly permanent, they are weak forces. For that reason, individual hydrogen bonds are not permanent and they are continually breaking and reforming as a consequence of the normal motion of molecules in solution.
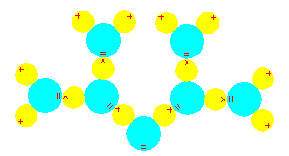
This illustration demonstrates how the positively charged side of one water molecule is attracted to the negatively charged side of another water molecule. In this way, the hydrogen atom is a bridge that links two oxygen atoms together, and the water molecules as well. This is the fundamental principle behind hydrogen bonds. In the case of water, many molecules can participate, and each of the hydrogen atoms (yellow) can link two oxygen atoms (light blue).
Reference
- Vinogradov, S. N. & Linnell, R. H.
Hydrogen Bonding
Van Nostrand Reinhold Co., NY, NY, USA





