Picric Acid
2,4,6-trinitrophenol
C6H3N3O7
Before You Begin
Please consult the following guide to safe working with this chemical fixing agent, including how to safely clean up spills.
Safety Note
Usually one of the first points made in any article about picric acid is that it can be explosive, or that it and its salts have been used as military high explosives before the introduction of TNT. That is all true. However, the dangers to histotechnologists from this are badly exaggerated, and picric acid can be used very safely in a histology laboratory. Its use should not be curtailed from a fear of an explosion, since the conditions under which that could take place are extremely unlikely to be present.
For an explosion to happen, the picric acid must be completely dry. Yet, picric acid is purchased wet with either water or ethanol, and as long as it remains in that state, it will not explode. In fact, since it is nearly always used as either a saturated aqueous solution or a saturated ethanolic solution, it makes sense to store it in two jars (one with an excess of distilled water and the other with an excess of absolute ethanol), thereby ensuring that both saturated solutions are always available. Simply replace the amount of saturated solution removed with distilled water or ethanol, as appropriate, after removing the amount required at the time. Following this practice ensures there is no danger of an explosion.
Instructions are sometimes encountered to weigh an amount of picric acid for incorporation into a solution. Since this involves handling a dry sample, technologists have become wary of doing so, but it was never the intention to completely dry picric acid for weighing, and in the past, this was understood. Histological solutions are rarely required to be accurate to anything more than one decimal place, and often not even that, so extreme accuracy is not needed. The usual practice was to place some wet picric acid onto a filter paper, then place the filter paper onto some other absorbent paper (newspaper, paper towels) for a few minutes to drain off the excess moisture. While still damp, the amount of picric acid required was weighed, but erring on the heavy side. This was then placed into the solvent being used and the papers rinsed and discarded. No completely dry picric acid was involved.
Even this practice is not usually necessary. Since picric acid is usually used in aqueous or ethanolic solution, it is simpler just to measure the required amount of picric acid by volume, then dilute as necessary. The volumes are based on the amount of picric acid in a saturated solution. Below are the figures (taken from the Merck Index) used on this site as the volume of a saturated solution that will contain one gram of picric acid at room temperature.
- Water: 1 gram in 78 mL
- Ethanol: 1 gram in 12 mL
Simply multiply the volume that contains one gram by the number of grams needed, and use that. For other solutions, such as those that use acetone, it is very unlikely that the small volume of ethanol used as a carrier for the acid would interfere with the staining reaction.
It is advisable to check the jars of picric acid periodically to ensure that dried acid is not crusted around the lid. This can be avoided by always wiping the edge of the jar to remove any drops of the solution before replacing the lid. If the seal under the lid is intact and in good condition, and the lid firmly tightened, there should be no evaporation or crusting. Similarly, ensure that wicking does not occur by never allowing a string or other absorbent material to be elevated above the surface of the picric acid, and never have it draped over the edge of the container. Doing so may enable the solution to wick up the material and crystallize on the end of it in a dry state.
Should some dried picric acid be encountered, do not panic. It is unlikely to explode. The easiest way to deal with such a problem is to fill a large bucket or sink with tap water, then place the crusted jar into it so that it sinks. The acid will then dissolve off. After an appropriate time, the jar may be removed and cleaned. The lid may then be undone and resealed properly.
If you are not comfortable with that, then involve your institution’s safety officers and have them deal with it.
Description
Picric acid is used as both a dye and as a fixing agent, and is a valued reagent in both cases.
When used as a fixing agent, it leaves tissues in a condition that makes them particularly receptive to staining with acid dyes, and methods such as Masson’s trichrome stain quite brilliantly as a result. This is so distinctive that many reference texts include in their instructions for this and other trichrome staining methods, a recommendation that sections from formalin fixed tissues be refixed in a picric acid fixative in order to improve the intensity of staining. The solution used is often Bouin’s fluid, although a simple saturated aqueous solution of picric acid is as effective and does not have the complication of giving off formalin fumes.
This improvement in staining does not extend to basic dyes, unfortunately, and results from using them may be disappointing. It should be noted that since picric acid is a fairly strong acid, it can hydrolyze DNA if tissues are left too long, and nuclear staining may be reduced. In some cases, a Feulgen stain for DNA may be positive without further hydrolysis in hydrochloric acid. For that reason, it should be avoided if nuclei are of particular interest as that method is unreliable on picric acid fixed material.
It leaves tissues in a reasonably soft state and sectioning is not usually impaired.
How it Fixes
Proteins
Picric acid is both an additive and precipitant fixing agent. It is believed that it forms picrates with basic amino acids and in the process causes the proteins to precipitate. It is suggested that this is the reason that acid dye staining is so effective following picric acid fixation: that basic proteins, to which acid dyes would attach, are well preserved, but acid proteins are not adequately fixed and may be removed. Nuclear protein is also precipitated, but the DNA itself remains water soluble. This means that nuclear structure may be shown with acid dyes rather than basic dyes, and that DNA methods are unreliable.
Carohydrates
There is no direct reaction with carbohydrates, although the protein component of carbohydrate-protein complexes may be fixed. Glycogen is unaffected directly but is either physically trapped within precipitated protein or, if it is bound to protein, is preserved along with the protein as it is fixed. This preservation of glycogen is striking enough that picric acid fixation is recommended when glycogen is of special interest, particularly if it is used in conjunction with high concentration of ethanol.
Lipids
Lipids are not affected, but the protein component of lipid-protein complexes may be fixed.
Morphology
Tissues tend to shrink. It is often stated that the glomeruli in mammalian kidney are so severly affected that picric acid fixation should never be employed for them. This may be somewhat overstated, and comparisons should be made if this is a concern. Formalin and other fixatives also causes shrinkage of glomeruli, yet are considered to be quite adequate for use. Compare the three photographs of a glomerulus below.
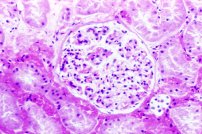
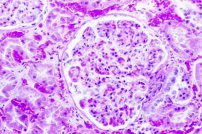
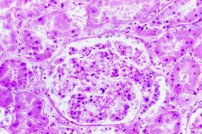
Time
Picric acid penetrates slowly but still a 3 mm to 5 mm thick piece of tissue will be fixed after about 8 hours, and certainly thoroughly fixed overnight. Tissues should not be left longer than necessary in the fixative due to the likelihood of acid hydrolysis of DNA.
Simple Solution
Except when sometimes used to secondary fix sections before trichrome staining, picric acid is never used alone. It is commonly mixed with ethanol, formalin and acetic acid and is considered compatible with most other fixing agents.
Aftertreatment
Conflicting advice is given for the aftertreatment of picric acid fixed tissues. This has arisen due to the assumption, often repeated, that picric acid forms protein picrates during fixation and that these protein picrates are water soluble. The advice given as a consequence is that picric acid fixed tissues should never be washed in water after fixation as it will cause some of the material to be washed out. This is quite overstated, and washing picric acid fixed tissues in running tap water appears to have little effect on the final morphology. It is recommended to test this out by fixing a 3 mm thick piece of mammalian kidney in Bouin’s fluid overnight, then washing in running tap water for three days before processing and staining. Compare to a similarly treated piece of tissue fixed with 10% NBF.
Following picric acid fixation, the tissue will be bright yellow from residual picric acid and many technologists want to remove this as they consider it to be a nuisance during processing, discoloring the dehydrating ethanols and clearing fluids. As it dissolves more efficiently in ethanol than water, it is more efficient to remove this discoloration with ethanols, so placing the tissues directly into 70% ethanol removes the yellow color more effectively than water washing.
Rather than wash extensively, some technologists briefly wash off the excess picric acid, then place the tissue in a dilute carbonate solution to more quickly eliminate the yellow discoloration by converting it to sodium or lithium picrate which quickly dissolves.
Other technologists do not consider the yellow color to be a problem and simply soak the tissue in 70% ethanol until it is time for routine processing to begin, then process normally. The tissue remains yellow and appears yellow in the block, thereby indicating the fixative used. If it interferes with staining, any residual yellow color may be removed by treating the sections with carbonate for a few minutes prior to staining. In most cases, it causes no problems, being replaced by the first acid dye applied.
References
- Baker, John R., (1958)
Principles of biological microtechnique
Methuen, London, UK. - Susan Budavari, Editor, (1996)
The Merck Index, Ed. 12
Merck & Co., Inc., Whitehouse Station, NJ, USA






