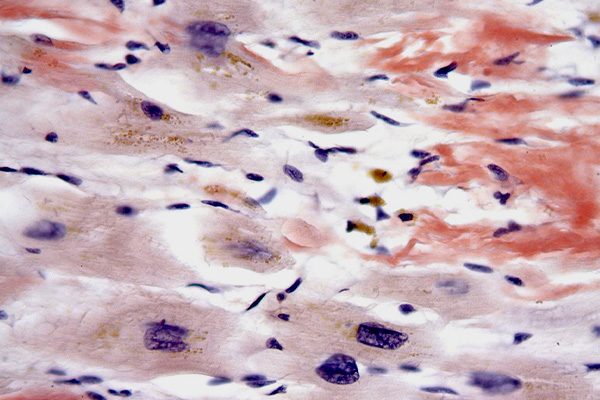
Akita & Kaneko's Hemalum
for Elastic Fibers
Materials
| Material | Amount | |
|---|---|---|
| Hematoxylin | 100 | mg |
| Ethanol, 70% | 100 | mL |
| Sodium iodate | 20 | mg |
| Potassium alum | 300 | mg |
| Chloral hydrate | 5 | g |
| Citric acid | 100 | mg |
Staining Solution Preparation
- Dissolve the hematoxylin into the ethanol.
- Add the potassium alum, then the other ingredients.
- Mix well for 15 to 20 minutes, then filter.
- At this point the solution is stable for 2 months at room temperature.
- Immediately prior to use, adjust to pH 8.0 with saturated aquueous lithium carbonate or 1N potassium hydroxide.
Tissue Sample
5µ paraffin sections of neutral buffered formalin fixed tissue are suitable. Helly and Bouin are also suitable. Other fixatives should be tested.
Protocol
- Bring sections to water via xylene and ethanol.
- Place into freshly pH adjusted staining solution for 30-60 minutes.
- Wash with running tap water for 10 minutes.
- Dehydrate with ethanols.
- Clear with xylene and mount with a resinous medium.
Expected Results
- Elastic fibres – blue
- Collagen – pale violet
Notes
- Note that this is an ethanolic variant of Mayer’s hemalum.
- If sodium iodate is left out, the staining solution lasts 3-4 days, but the staining time is extended to 60 minutes.
- The solution may also be made by diluting 30 mL Mayer’s hemalum with 70 mL absolute ethanol, mixing for 20 minutes, and filtering before adjusting the pH. Staining requires 60 minutes and the results are paler.
Safety Note
Prior to handling any chemical, consult the Safety Data Sheet (SDS) for proper handling and safety precautions.
References
- Akita, M. and Kaneko, K., (1981)
An improved hematoxylin method with application of Mayer’s hemalum for staining of elastic fibres.
Stain Technology, v. 56, p. 327 - Kaneko, K. and Akita, M., (1978)
On the staining of elastic fibres with alum hematoxylin.
Stain Technology, v. 53, p. 43.