Lewis and Miller's Trichrome
for Pituitary cells
This is a modification of Kricheski’s method.
Materials
Solution A
| Material | Amount | |
|---|---|---|
| Acid fuchsin | 0.25 | g |
| Distilled water | 100 | mL |
Solution B
| Material | Amount | |
|---|---|---|
| Methyl blue, 1% aqueous | 30 | mL |
| Orange G, 1% aqueous | 30 | mL |
| Phosphomolybdic acid, 1% aqueous | 30 | mL |
Tissue Sample
5µ paraffin sections of neutral buffered formalin fixed tissue are likely suitable. Other fixatives are also likely to be satisfactory. Most trichrome stains benefit from picric acid or mercuric chloride fixation. Formalin fixed tissues may benefit from secondary fixation of sections in Bouin’s fluid.
Protocol
- Bring sections to water via xylene and ethanol.
- Place into solution A for 30 minute.
- Rinse well with distilled water.
- Place into solution B for 24 hours.
- Dip 2 or 3 times in 70% ethanol.
- Dehydrate and differentiate with ethanol for 1-3 minutes.
- Clear with xylene and mount with a resinous medium.
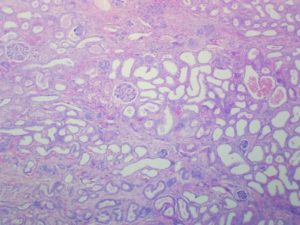
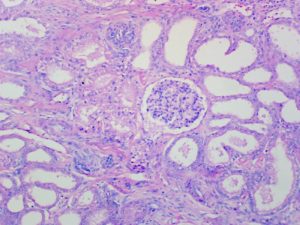
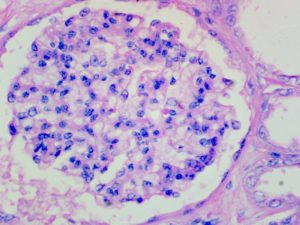
Expected Results
- Nuclei – red
- Acidophils – orange red
- Basophils – blue
- Chromophobes – Pale grey
Notes
- Although not specified, an acid resistant nuclear stain such as Weigert’s iron hematoxylin could be inserted prior to staining with solution A.
Safety Note
Prior to handling any chemical, consult the Safety Data Sheet (SDS) for proper handling and safety precautions.
References
- Gray, Peter. (1954)
The Microtomist’s Formulary and Guide.
Originally published by: The Blakiston Co.
Republished by: Robert E. Krieger Publishing Co.
Citing:
Kricheski, (1931)
Stain technology, v.6, pp.97
And:
Lewis and Miller, (1938)
Stain technology, v.14, pp.111