Dyes may be structured according to their structure, color index, or other alternative means.
Acridine
Acridine dyes are derived from acridine:
They have the general formula:
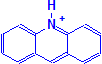
The general formula shows the chromophore to be a quinoid ring. It should also be noted that the molecule is positively charged, shown on the nitrogen atom attached to the quinoid ring, and the dyes are consequently basic. There are a few dyes in this class that are used in histotechnology, but they tend to be for fairly specialized purposes which take advantage of their fluorescent characteristics. The commonest examples would be acridine orange and acriflavine.
Anthraquinone
Anthaquinone dyes are derived from anthracene:
They have the general formula:
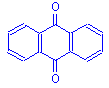
The general formula shows the chromophore to be a quinoid ring. This class of dyes may have either hydroxyl groups or amino groups attached to the general structure. Those that have hydroxyl groups (hydroxyanthraquinones) are the more numerous for histological purposes, and include the mordant dyes alizarin and alizarin red S, both used to demonstrate calcium. Also represented is anthracene blue SWR, a substitute for hematoxylin. Dyes having amino groups (aminoanthraquinones) are less common, but include nuclear fast red, a mordant dye useful for staining nuclei red.
Arylmethane
Arylmethane dyes are derived from methane:
Diarylmethanes have the general formula:
Triarylmethanes have the general formula:
Arylmethane dyes are so called because they are derived from methane, but some of the hydrogen atoms are replaced with aryl rings. Since a synonym for aryl is phenyl, they are also referred to phenylmethane dyes. Aryl rings are often referred to as benzene rings. However, benzene is a specific chemical compound and this usage is technically incorrect, although very common.
Diarylmethanes
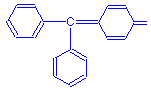
Diarylmethane (or diphenylmethane) dyes have two aryl rings. Only one dye in this subgroup has gained much use, and that is auramine O, a fluorescent dye used for the demonstration of acid fast bacteria and to make a fluorescent Schiff reagent. Its structural formula is shown below. Note the alkylamino groups attached to the aryl rings. This dye is subclassified as an aminodiarylmethane as a consequence. The chromophore is the carbon to nitrogen double bond.
Triarylmethanes
Triarylmethane (or triphenylmethane) dyes contain three aryl rings. These are much more common and numerous examples can be given. The pararosanin homologues are good examples. Pararosanilin itself is used to make Schiff’s reagent, and basic fuchsin is used to make carbol fuchsin, important for the identification of tuberculosis. These dyes also contain amino groups, and for that reason are sometimes identified as aminotriarylmethane dyes, and are often basic. In pararosanilin, shown below, each of the three aryl rings has an amino group. The quinoid ring is the chromophore.
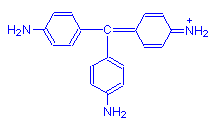
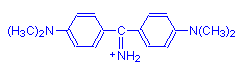
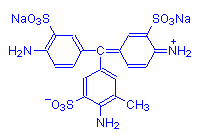
Not all aminotriarylmethane dyes are basic. Some are acid dyes. These have strongly anionic sulphonic groups as well, making the overall charge on the molecule negative. Examples are acid fuchsin (shown below) and methyl blue.
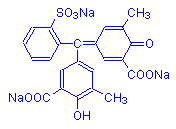
Another subgroup of the arylmethanes has hydroxyl groups instead of amino groups. These dyes are termed hydroxytriarylmethane dyes, and are acid dyes. An example is mordant blue 3, shown below, which has been recommended as a hematoxylin substitute. Dyes in this subgroup are not widely used.
There are other subgroups of arylmethane dyes, but they are rarely used in histotechnology.
Azo
Azo dyes contain the azo chromophore:
They have the general formula:
Azo dyes are subgrouped according to the number of azo chromophores in the molecule.
Monoazo
Disazo
Trisazo
Trisazo dyes contain three azo chromophores. These are not common in histotechnology.
Tetrakisazo
Lysochromes
Lysochromes, or fat dyes, are usually disazo dyes and frequently have a single hydroxyl group ortho to the azo nitrogen. Lillie, in Conn’s Biological Stains, draws attention to the work of Michaelis in the early part of the 20th century, saying that when this is so, intramolecular reconfiguration occurs which results in the loss of hydrogen from the hydroxyl, gaining of a hydrogen by one of the azo nitrogens, and formation of an ortho quinoid ring. While still intensely colored, the resulting compound is insoluble in water, but is soluble in non-polar solvents, including triglycerides.
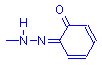
Diazonium
Diazo compounds are derived from a primary arylamine:
Following treatment with nitrous acid (HNO2), the amino group is converted to an azo group:
Diazonium compounds have the general formula:
They are more commonly represented in the charged state by:
Burstone describes diazo compounds as containing:
“The N2 grouping combined to only one hydrocarbon radical instead of two as in azo compounds. The formation of diazo compounds depends upon the replacement of three hydrogen atoms of the salt of an aryl primary amine by nitrogen from nitrous acid to form the so-called diazonium salts.”
Examples are:
Due to their instability, commercially available diazonium compounds may not be supplied in the form shown above, but as some other complex. Burstone gives the following possibilities:
- Diazonium chlorides, double salts with zinc chloride (or manganese, cobalt or cadmium chlorides)
- Diazonium acid sulphates
- Diazonium alkyl sulphates
- Diazonium chlorides
- Diazonium fluoborates
- Diazonium benzenesulphonates
- Diazonium acid 1,5-naphthalenedisulphonates
Strictly speaking, diazo compounds are not dyes, but dye precursors. Some azo dyes are manufactured by coupling a phenol or naphthol to a diazonium compound.
In histotechnology, they have two main uses. The first is for the demonstration of enterochromaffin, traditionally using fast red B although many others will do so. The second is for demonstrating the activity of some enzymes, for example alkaline and acid phosphatases. Using naphthol phosphates as the substrate, these enzymes split off the phosphate, leaving the naphthol, which then couples to the diazo compound to form an insoluble dye at the site of activity. Similar processes are used for other enzymes as well.
In all three uses the reaction is fundamentally the same. The differences are that when using azo coupling to manufacture dyes, the naphthol used would have appropriate groups to ensure a soluble product. To demonstrate enzyme activity, an insoluble product is needed so that it can be precipitated where the enzyme is. Enterochromaffin, being a tissue component, stays where it is with the new dye attached.
Diazo compound
+
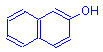
β-naphthol
=
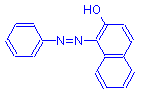
Azo dye
+ HCl
The azo dye in the above example has a hydroxyl group ortho to an azo nitrogen. Under these circumstances, there is some molecular rearrangement, analogous to that of lysochromes, causing the dye to become insoluble.
Azo coupling is not limited to naphthols. Burstone gives the following list of compounds capable of being coupling components with diazonium compounds:
- compounds with phenolic hydroxyl groups, naphthols and analogues
- primary (and some secondary) arylamines
- aromatic compounds with a reactive methylene group capable of enolization, including derivatives of acetoacetic ester and arylamines
- some heterocycles such as pyrrole, indole and imidazole
- o-alkylphenols, naphthols and N-acylamines, including phenol and naphthol ethyl ethers and acetylnaphthylamines
- aliphatic enols such as ethyl acetoacetate, and compounds containing an alicyclic carbon such as ascorbic acid or dimedone
Nitro
Nitro dyes contain the nitro chromophore:
NO2
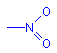
They have the general formula:
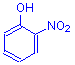
The empirical way of designating the nitro group does not indicate that its electrons are delocalized. The bonds to the two oxygen atoms are of equal strength, and are shown as a single and a double bond only for convenience. The electrons from both bonds are shared, and these delocalized electrons are integrated with the delocalized electrons of the aryl rings of dyes, causing absorption in the visible range. The dyes so formed are invariably acid dyes. Picric acid and martius yellow are the usual examples.
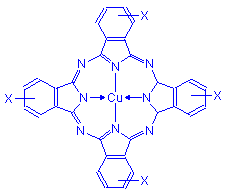
Phthalocyanine
The phthalocyanine dyes are few in number, but those few are commonly used. Alcian blue is employed in one of the standard methods for acid mucopolysaccharides. Luxol fast blue is used to demonstrate myelin.
Phthalocyanin dyes have the general formula below. These vary with each dye. At the point marked X various groups are attached.
Quinone-imine
Quinone-imine dyes are based on the structure of the fictional compound para-quinone-di-imine, from which the name of the dye class originates.
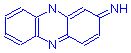
There are several subgroups of quinone-imine dyes. Three are important in histotechnology, two are not. The two unimportant subgroups are the indamins and the indophenols. The three important subgroups are the azins, the oxazins and the thiazins, incorporating 2 nitrogens, nitrogen and oxygen, and nitrogen and sulphur atoms, respectively.
Azins have the general formula shown below. The group is further subdivided into three subgroups: eurhodins, safranins and indulins. The eurhodins are the simplest, with attached amino groups, illustrated by neutral red, a dye of considerable importance. Similarly, the safranins, which have an aryl ring attached to a central nitrogen, include safranin O. The indulins are not commonly used, but nigrosin has value for negative staining.
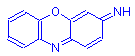
Oxazins have the general formula shown below. The oxazins include gallocyanin, gallamin blue and celestin blue B. These three dyes have been recommended as hematoxylin substitutes in H&E staining when used in solution with iron alum.
Thiazins have the general formula shown below. Thiazins are particularly important as they include several very commonly used dyes, including some that are strongly metachromatic. These include the methylene blue homologues.
Tetrazolium
Tetrazolium compounds have the general formula:
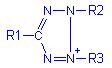
An insoluble colored formazan results on reduction. These have the general formula:
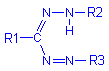
Strictly speaking, tetrazolium compounds and their formazans are not dyes. Their characteristic structure is the five-member ring of one carbon and four nitrogens atoms. They may contain one or two of these structures. Examples are iodonitrotetrazolium and nitro blue tetrazolium.
When this characteristic structure is reduced by the addition of a hydrogen atom, the nitrogen-to-nitrogen bond is broken with the formation of the corresponding formazan. Formazans are intensely colored but water insoluble, so they precipitate as they form. They are valuable for the demonstration of enzymes that transport hydrogen, such as dehydrogenases, where they act as hydrogen acceptors.
Thiazole
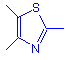
Thiazole dyes contain the thiazole ring:
They have the general formula:
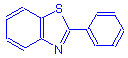
Thiazole dyes are not well represented in histotechnology. The class does, however, include thioflavine T, a valuable dye for the fluorescent demonstration of amyloid. The indamine group (–N=) is the chromophore.
Xanthene
Xanthene dyes are derived from xanthene. This class of dyes is divided into three subgroups: Fluorenes, fluorones and rhodols. Fluorenes and fluorones contain dyes of importance in histotechnology, the rhodols do not. The fluorenes are further subdivided into five groups. Of these the pyronins and rhodamines include dyes we use. The others do not.
The pyronin subgroup of fluorenes have the general formula shown to the right. The dyes pyronin Y and pyronin B are examples.
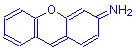
The rhodamine subgroup of fluorenes have the general formula shown to the right. Rhodamine B is an example.
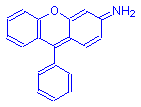
Fluorone Dyes
Fluorones have the general formula shown to the right. The fluorones are sometimes referred to as the eosins, and include many dyes used as counterstains to alum hematoxylin. Their general formula shown above is actually that of fluorescien, from which they are derived.

References
- R. D. Lillie.
Conn’s Biological Stains
Williams & Wilkins, Baltimore, MD., U.S.A. - Michaelis, L.
Ueber fett-farbstoffe,
Virchow. Arch. Path. Anat., v.164, 263-270 (1901). - Burstone, M.S.
Enzyme Histochemistry and its application to the study of neoplasms
Academic Press, New York, NY, USA





